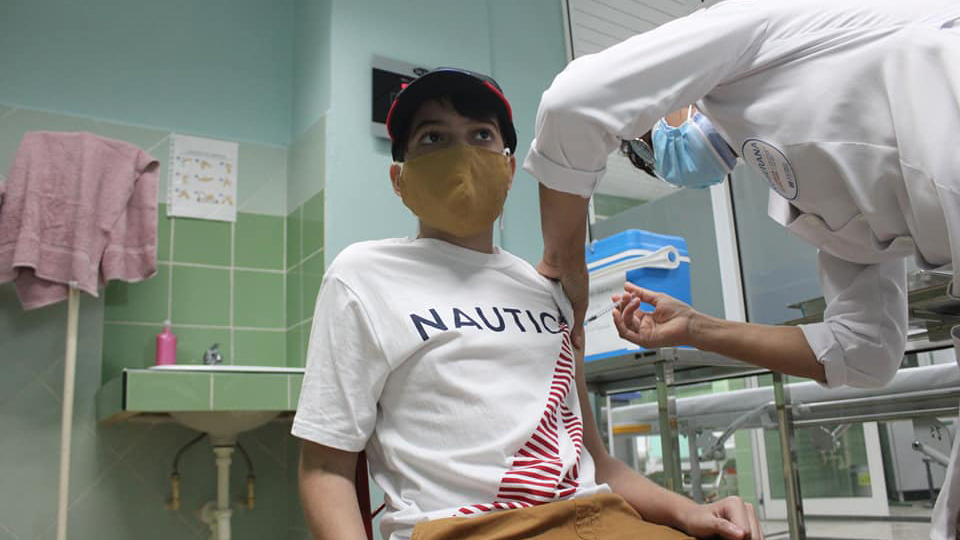
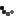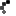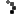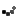Cuba established its scientific prowess during COVID-19 by inventing not one, but five vaccines to contain the virus. All vaccines were developed in the public sector and demonstrated the importance of sharing and collaboration in scientific research. Three of the vaccines, which were approved for use, have proven to be safe and have nearly 90% efficacy, among the highest in the world.
The Cuban government has been collaborating with other countries to expand production of vaccines. Iran was the first country to begin producing Soberana-2, a conjugate vaccine developed by Finlay Institute of Vaccines (IFV), Havana. This was a much needed collaboration in 2021 as both countries face heavy sanctions by the US and as a result were facing an acute shortage of vaccines.
Italy is the latest to join in that collaboration. A memorandum of understanding was signed between IFV, Italian company ADIENNE Pharma & Biotech and the Italian Agency for Economic and Cultural Exchange with Cuba (AICEC) in April 2022. The Italian company will begin production of Soberana-2 soon in Italy, which will become part of an international health cooperation mechanism and be exported to other countries.
“We have reached this agreement, moved in this direction, which is a very positive move for our vaccine,” said Vicente Vérez, general director of the IFV.
“This collaboration is a political initiative of great importance. It shows that science coming from Cuba is of value and the products are of very good standard. A European country doing this helps make space for Cuban products in the western nations. The vaccine, which has been developed in the public sector and has a public patent, should not be limited to a few million people,” said Nicoletta Dentico, Head, Global Health Program, Society for International Development (SID).
Overcoming the WHO PQ hurdle
The World Health Organization is the nodal international agency that approves vaccines, medicines and other medical tools for use. While countries such as the United States, Brazil, United Kingdom and India have a robust mechanism to analyze scientific and legal aspects of a medical product, most countries depend on the WHO for approval, through the WHO prequalification (WHO PQ).
Unfortunately, WHO has yet not approved the Cuban vaccines. According to scientists who spoke to People’s Health Dispatch, this is due to discriminatory regulatory practices adopted by the WHO. “Good Manufacturing Practices (GMP) is an important part of regulation. However, it focuses more on the manufacturing facility rather than the quality of the product,” commented a scientist adding that facilities in developing countries such as Cuba lose out due to such regulatory demands.
Collaborating with ADIENNE may help overcome this limitation. If ADIENNE’s manufacturing facilities secure GMP approval, then Soberana-2 may also achieve WHO PQ, a big step in exporting the vaccine to other countries.
However, it may not be that easy. Steven Solomon, Principal Legal Officer, World Health Organization (WHO), in a recent public event spoke in favor of mRNA vaccines and emphasized the importance of promoting them. Most of the mRNA vaccines fall under the monopoly of big pharmaceutical companies based in high-income countries. This bias against vaccines that originate from the Global South impacts the WHO’s decision making.
Dentico hopes that production in Italy will help. “This collaboration provides additional arguments and evidence that the West needs to psychologically accept vaccines from Cuba. It should convince the WHO to look at their value and understand the public health importance of Soberana 2. WHO should get engaged in promoting this vaccine as much as it does for the mRNA vaccines,” she said.
According to Vérez, the strategy is to continue pursuing WHO PQ for Soberana-2, as well as worldwide distribution of the vaccine, even in Europe and North America. “This requires alliances to share science,” he said.
The importance of Soberana-2
Soberana-2 was created to immunize children, unlike any other vaccine in the world. Through the technology of a conjugate vaccine which has historically proven to be effective among children, Cuba became the first country in the world to vaccinate children as young as 2 years old. This proved to be very effective in containing the spread of the Omicron variant in Cuba. “When mass vaccination is the main strategy, then it is important to vaccinate 60-70% of the population to achieve herd immunity. That cannot happen without vaccinating children,” said Prof. Fabrizio Chiodo, scientist at the Italian National Research Council (CNR) and a collaborator with IFV.
Production of Soberana-2 by ADIENNE will be beneficial for children in Italy and other countries where it is exported. “Based on an evaluation of the Soberana-2 vaccine itself and the impact that this immunogen has had on pediatric vaccination, we have managed to arouse the interest of this company, capable of producing the drug, thanks to the AICEC that has helped this approach,” said Vérez.
The history of Cuba-Italy collaboration during COVID-19
Cuba and Italy share a rich history of collaboration during the pandemic. Early on in the COVID-19 pandemic, Italy had some of the highest infection and mortality rates in the world. As its health system crumbled under the pressure of increased caseload, the Cuban government sent medical teams to some of Italy’s worst-hit regions, including Lombardy and Piedmont, to replace overworked Italian professionals.
“Our doctors got there when it was still unknown what could happen. Then we carried out the Soberana Plus-Turin clinical trial, in which Italian citizens came to Cuba to be vaccinated with Soberana Plus,” said García Rivera, Research Director, IFV. He was referring to the clinical trial of 38 Italians who took booster doses of Cuba’s other vaccine, Soberana Plus. He added that the signing of this memorandum is a third important action, “in which we are already, specifically, signing an agreement to start the production of the Soberana-2 vaccine in the final stages in Italy.”
Read more articles from the latest edition of the People’s Health Dispatch and subscribe to the newsletter here.